Latest Updates
-
 International Human Solidarity Day 2025: History, Significance, and Why It Matters
International Human Solidarity Day 2025: History, Significance, and Why It Matters -
 Purported Video of Muslim Mob Lynching & Hanging Hindu Youth In Bangladesh Shocks Internet
Purported Video of Muslim Mob Lynching & Hanging Hindu Youth In Bangladesh Shocks Internet -
 A Hotel on Wheels: Bihar Rolls Out Its First Luxury Caravan Buses
A Hotel on Wheels: Bihar Rolls Out Its First Luxury Caravan Buses -
 Bharti Singh-Haarsh Limbachiyaa Welcome Second Child, Gender: Couple Welcome Their Second Baby, Duo Overjoyed - Report | Bharti Singh Gives Birth To Second Baby Boy | Gender Of Bharti Singh Haarsh Limbachiyaa Second Baby
Bharti Singh-Haarsh Limbachiyaa Welcome Second Child, Gender: Couple Welcome Their Second Baby, Duo Overjoyed - Report | Bharti Singh Gives Birth To Second Baby Boy | Gender Of Bharti Singh Haarsh Limbachiyaa Second Baby -
 Bharti Singh Welcomes Second Son: Joyous News for the Comedian and Her Family
Bharti Singh Welcomes Second Son: Joyous News for the Comedian and Her Family -
 Gold & Silver Rates Today in India: 22K, 24K, 18K & MCX Prices Fall After Continuous Rally; Check Latest Gold Rates in Chennai, Mumbai, Bangalore, Hyderabad, Ahmedabad & Other Cities on 19 December
Gold & Silver Rates Today in India: 22K, 24K, 18K & MCX Prices Fall After Continuous Rally; Check Latest Gold Rates in Chennai, Mumbai, Bangalore, Hyderabad, Ahmedabad & Other Cities on 19 December -
 Nick Jonas Dancing to Dhurandhar’s “Shararat” Song Goes Viral
Nick Jonas Dancing to Dhurandhar’s “Shararat” Song Goes Viral -
 From Consciousness To Cosmos: Understanding Reality Through The Vedic Lens
From Consciousness To Cosmos: Understanding Reality Through The Vedic Lens -
 The Sunscreen Confusion: Expert Explains How to Choose What Actually Works in Indian Weather
The Sunscreen Confusion: Expert Explains How to Choose What Actually Works in Indian Weather -
 On Goa Liberation Day 2025, A Look At How Freedom Shaped Goa Into A Celebrity-Favourite Retreat
On Goa Liberation Day 2025, A Look At How Freedom Shaped Goa Into A Celebrity-Favourite Retreat
Serum Institute Seeks DCGI Nod For COVID Vaccine Covovax
The Serum Institute of India on Friday sought permission from the Drugs Controller General of India (DCGI) to manufacture COVID-19 vaccine Covovax for restricted use in emergency situations at its Manjari manufacturing site, official sources said.
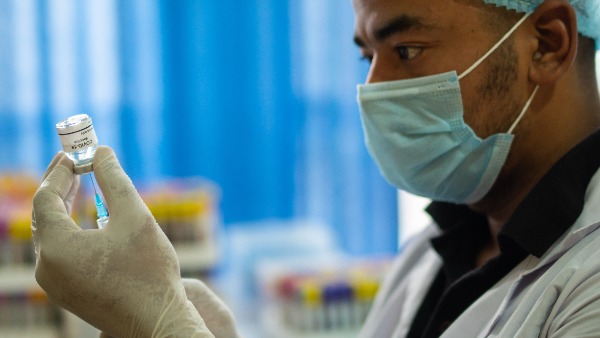
The company has submitted an interim phase 2/3 clinical study report on Indian adults, the interim report of Novavax UK and USA-Mexico phase-3 clinical study and response to the queries raised by the DCGI office, along with its application.
There were no safety concerns arising from the study data and Covovax is safe and immunogenic in the adult population, Prakash Kumar Singh, Director, Government and Regulatory Affairs at the Serum Institute of India (SII), is learnt to have said in the application sent to the DCGI.
"In our government's endeavour to fight against COVID-19 pandemic, we have also been working shoulder to shoulder with the Government of India to make available one more safe and efficacious world-class COVID-19 vaccine for our country and the world at large."
"Approval of our COVOVAX and its availability will further strengthen India's capability to fight COVID-19 pandemic and ensure vaccine security in line with our prime minister's clarion call of Atmanirbhar Bharat," an official source quoted Singh as having said in the application.
In the phase 2/3 study in India, more than 1,400 participants have received at least the first dose of the vaccine with no safety concerns reported so far, stated the application.
In August 2020, US-based vaccine maker Novavax Inc had announced a licence agreement with SII for the development and commercialisation of NVX-CoV2373, its COVID-19 vaccine candidate, in low and middle-income countries and India.
SII, which manufactures Covishield, plans to produce Covovax at its Manjari plant in Maharashtra.
India has given emergency use permission to several COVID-19 vaccines, including Covishield, Covaxin of Bharat Biotech and Russian-made Sputnik V.
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications












