Latest Updates
-
 Purported Video of Muslim Mob Lynching & Hanging Hindu Youth In Bangladesh Shocks Internet
Purported Video of Muslim Mob Lynching & Hanging Hindu Youth In Bangladesh Shocks Internet -
 A Hotel on Wheels: Bihar Rolls Out Its First Luxury Caravan Buses
A Hotel on Wheels: Bihar Rolls Out Its First Luxury Caravan Buses -
 Bharti Singh-Haarsh Limbachiyaa Welcome Second Child, Gender: Couple Welcome Their Second Baby, Duo Overjoyed - Report | Bharti Singh Gives Birth To Second Baby Boy | Gender Of Bharti Singh Haarsh Limbachiyaa Second Baby
Bharti Singh-Haarsh Limbachiyaa Welcome Second Child, Gender: Couple Welcome Their Second Baby, Duo Overjoyed - Report | Bharti Singh Gives Birth To Second Baby Boy | Gender Of Bharti Singh Haarsh Limbachiyaa Second Baby -
 Bharti Singh Welcomes Second Son: Joyous News for the Comedian and Her Family
Bharti Singh Welcomes Second Son: Joyous News for the Comedian and Her Family -
 Gold & Silver Rates Today in India: 22K, 24K, 18K & MCX Prices Fall After Continuous Rally; Check Latest Gold Rates in Chennai, Mumbai, Bangalore, Hyderabad, Ahmedabad & Other Cities on 19 December
Gold & Silver Rates Today in India: 22K, 24K, 18K & MCX Prices Fall After Continuous Rally; Check Latest Gold Rates in Chennai, Mumbai, Bangalore, Hyderabad, Ahmedabad & Other Cities on 19 December -
 Nick Jonas Dancing to Dhurandhar’s “Shararat” Song Goes Viral
Nick Jonas Dancing to Dhurandhar’s “Shararat” Song Goes Viral -
 From Consciousness To Cosmos: Understanding Reality Through The Vedic Lens
From Consciousness To Cosmos: Understanding Reality Through The Vedic Lens -
 The Sunscreen Confusion: Expert Explains How to Choose What Actually Works in Indian Weather
The Sunscreen Confusion: Expert Explains How to Choose What Actually Works in Indian Weather -
 On Goa Liberation Day 2025, A Look At How Freedom Shaped Goa Into A Celebrity-Favourite Retreat
On Goa Liberation Day 2025, A Look At How Freedom Shaped Goa Into A Celebrity-Favourite Retreat -
 Daily Horoscope, Dec 19, 2025: Libra to Pisces; Astrological Prediction for all Zodiac Signs
Daily Horoscope, Dec 19, 2025: Libra to Pisces; Astrological Prediction for all Zodiac Signs
Bharat Biotech Submits Clinical Trial Data Of Covaxin In Children To CDSCO
Bharat Biotech, which completed the Phase 2/3 trials of Covid-19 vaccine Covaxin for use in children under 18 years of age, has submitted the data to the Central Drugs Standard Control Organisation for its verification and subsequent approval for Emergency Use Authorisation for the jab, company sources said on Wednesday.
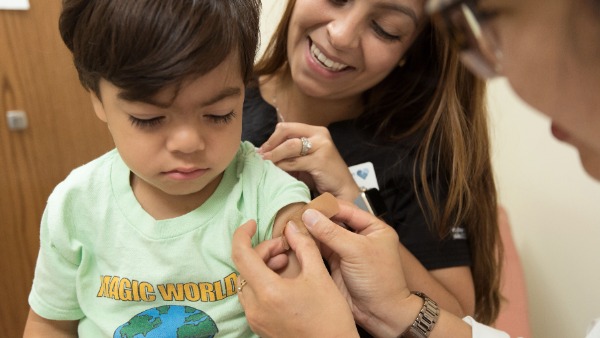
"Covaxin clinical trial data of 2-18 years age group has been submitted to CDSCO. This is feasible due to the safety of the manufacturing platform and empirical evidence from Phase 1,2 and 3 clinical trials in adults," sources told PTI.
Chairman and Managing Director of Bharat Biotech International Ltd, Krishna Ella, had said on September 21 that the Paediatric Covaxin completed phase 2/3 trials with about 1,000 subjects and the data analysis was going on.
As part of the Phase II/III trial, the two-dose Covaxin was administered with 28 days apart.
"We will be submitting the data (to the regulator) by next week," he had said.
He also had said that Phase 2 trials of an intranasal vaccine to prevent Covid-19 were going on and expected to be over in October.
If approved, Covaxin will be the first Covid-19 vaccine that can be administered to children in India.
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications












